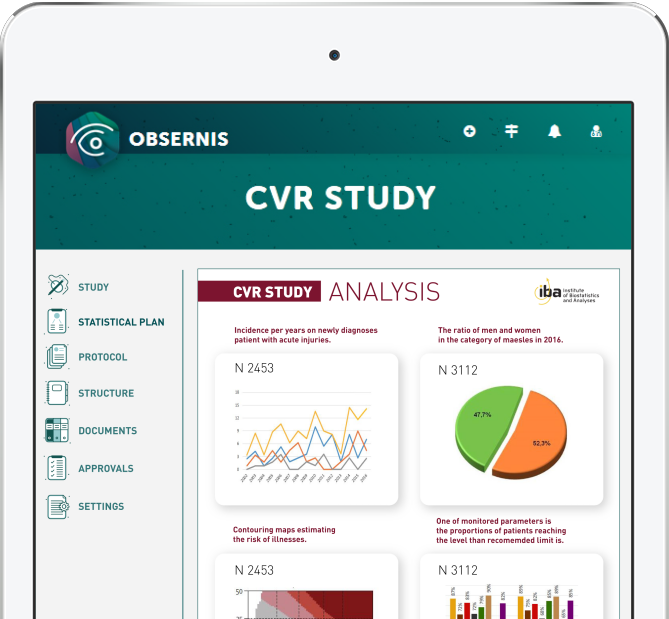
OBSERNIS is your electronic Trial Master File
Simple Guide For Observational/Non-Interventional Studies.
OBSERNIS is a unique and innovative approach to setting up a non-interventional or observational study. We have carried out a detailed research of the definition of the type of study and we have found that there is no simple unification.
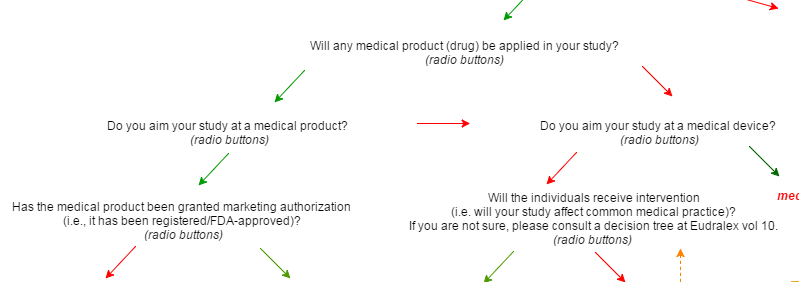
As a result, we have developed a special algorithm that can help you define a study type according to EU and US legislative regulations with a simple wizard.
Key Features
-
Study type definition
A set of simple questions can help you define your study type.
-
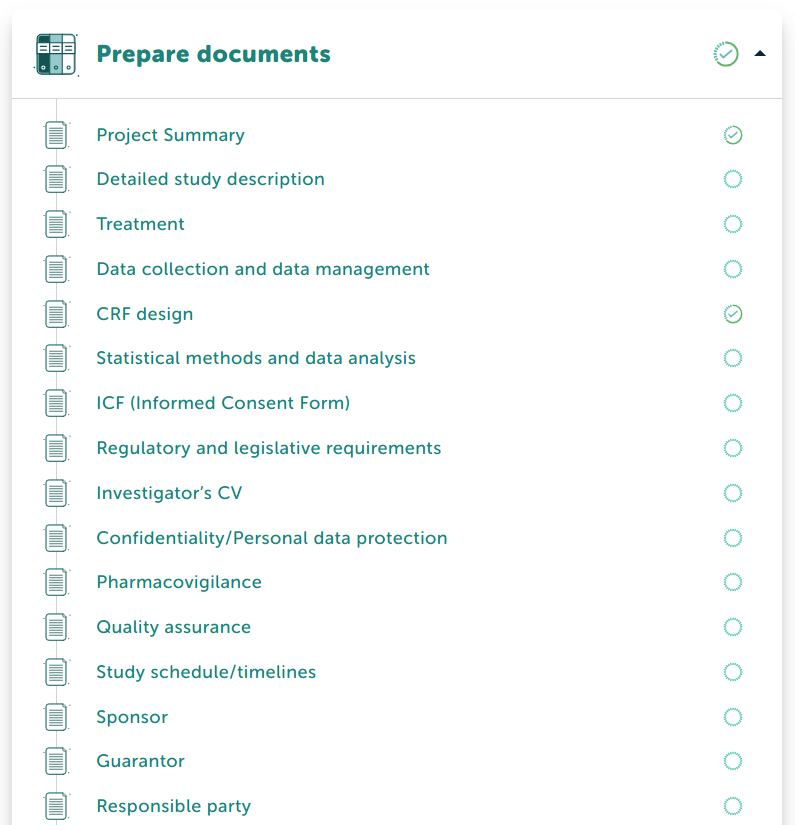
Documents management
Helps you create study documents on spot or upload your own documents.
-
Trial Master files on cloud
You have tessential documents in one place. Anytime. Simply share with your colleagues.
-
Safe your budget
You can buy any part of our services or order a specific consultation.
-
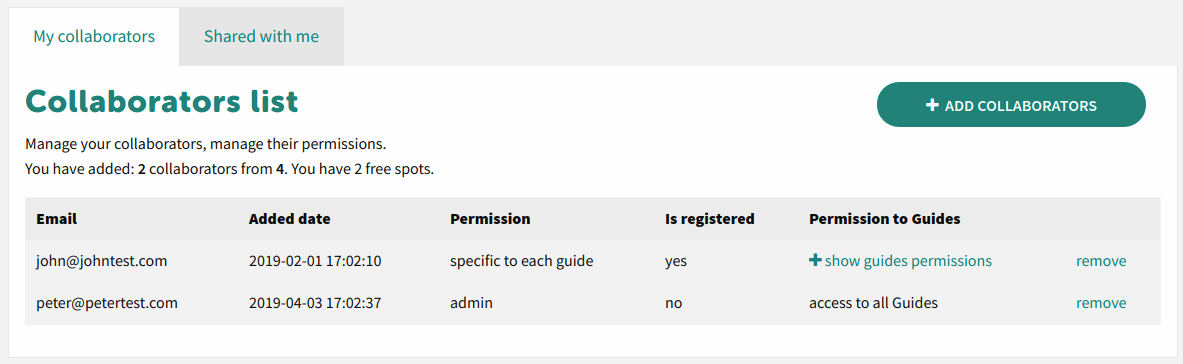
Collaboration with colleagues
You can share easily project with your colleagues through specific rules or share with anybody for reading.
-
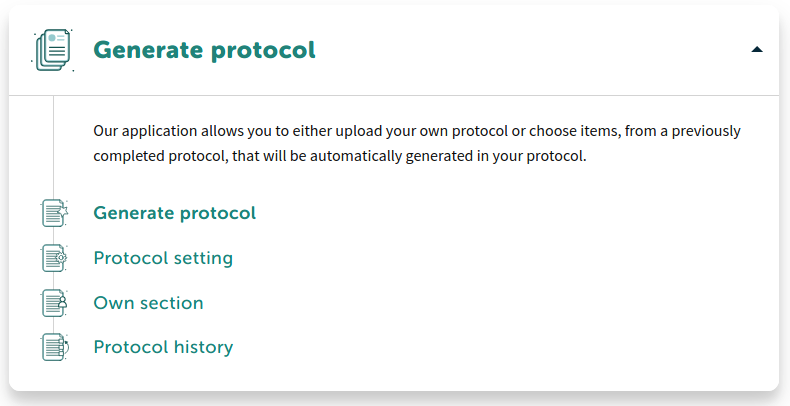
Generate Protocol
Define your structure, language, chapters easily and generate your specific protocol for clinical study..
-
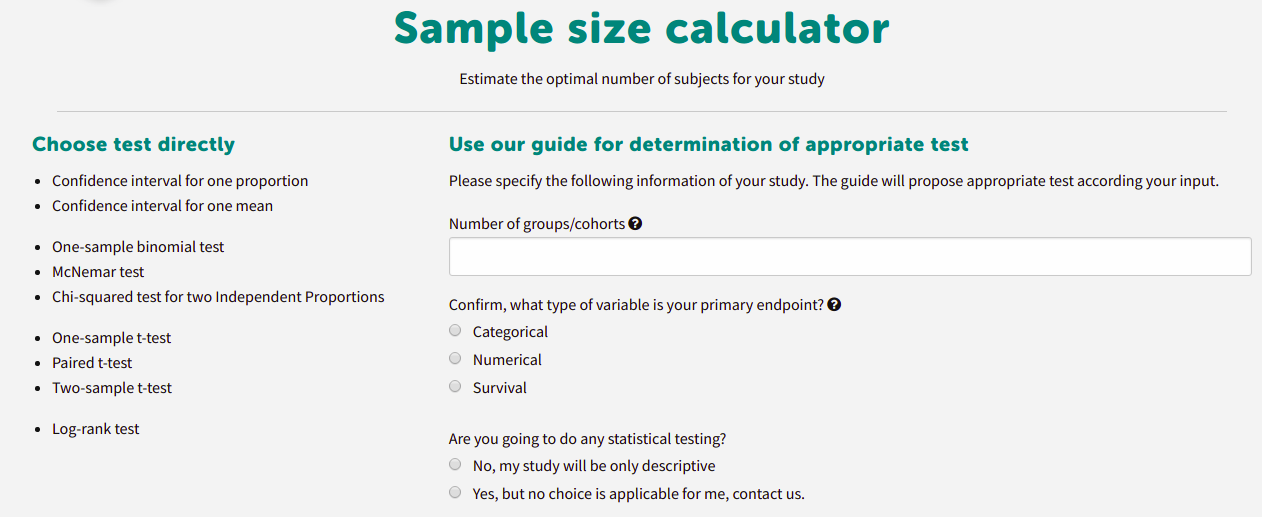
Calculate your Sample size
No need to have special statistical software. You can generate Sample size automatically.
-
Legislation compliance
No need to go through heavy legislation, we have done that for you.
Pricing
We support science and research, and we are bringing a solution that will help simplify the administrative burden at a unique price. We are pleased with each new healthcare project. Your Healthy Data.
Free
1 project- Study definition wizzard
- Trial master file
- Document upload
- Protocol generation
Professional
Unlimited number of projects- Study definition wizzard
- Trial master file
- Document upload
- Protocol generation
- Team sharing
- Own logo
- Own chapters
- SAP automated
billed annually

About us
We are Institute of Biostatistics and Analyses Ltd, a spin-off company of the Masaryk University.
By the years of experience with managing clinical research projects and helping to set them up, we have noticed how the cumbersome procedures in the projects wrinkle up our client’s foreheads. As we also focus on computer automation, we have decided to iron them out by releasing ObserNIS as our brand new product, hence sharing some of our hardly-gained know-how with those seeking for help. If you want to know more about us, please refer to our website.

-
Real world evidence
We breathe life into research based on real world data.
-
Over 10-year
experience in the fieldFor more than a decade we have been dealing with observational/non-interventional studies, EHRs, registries, etc.
-
Compliant
ISO/IEC 20000-1:2006, EN ISO
ČSN EN ISO 9001:2001/2009, ISO/IEC
27001:2006.